Oh No!
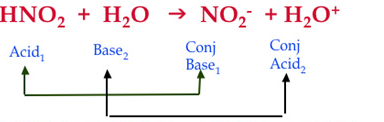
In the equation above you can see the acid (HNO2) reacting with the base (H2O).
To figure out the conjugate acid, the products to the right must be examined.
The acid loses a hydrogen ion to create NO2 ; therefor NO2 must be the conjugate base.
The base receives a hydrogen ion to create H2O ; therefor H2O must be the conjugate acid
To figure out the conjugate acid, the products to the right must be examined.
The acid loses a hydrogen ion to create NO2 ; therefor NO2 must be the conjugate base.
The base receives a hydrogen ion to create H2O ; therefor H2O must be the conjugate acid